Our Research
We are working to understand how cells respond to viral pathogens, and how these signaling pathways can be harnessed for new potential therapies to treat cancer and autoimmune diseases. Importantly, many aspects of the cellular response to viral infection remain unknown. Our lab uses an approach where we seek to reconstitute signaling outside of the cell using highly purified components to understand the mechanisms that control human immunity.
Nucleotide Immune Signaling
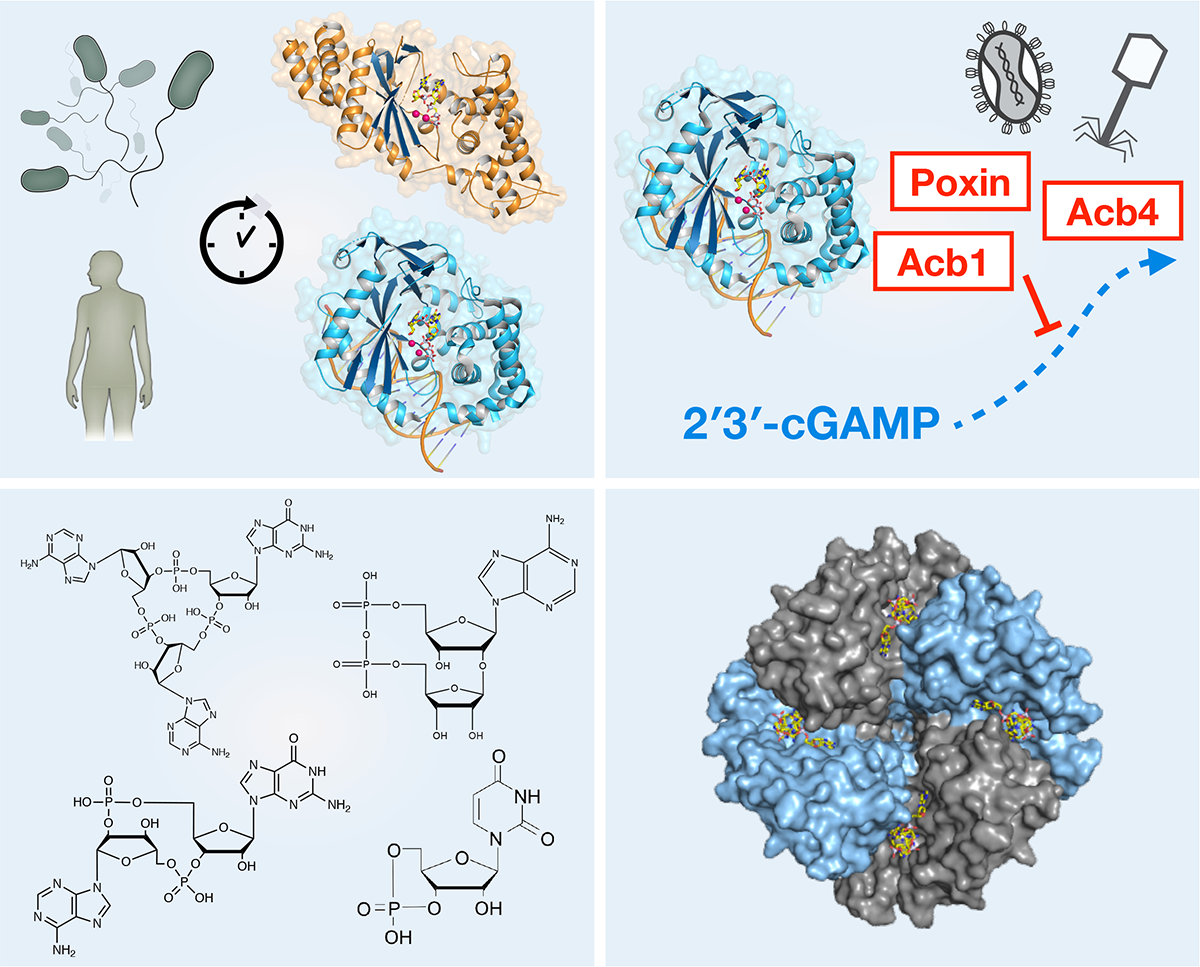
Nucleotide immune signals are specialized molecules that allow cells to activate immunity and control the response to pathogen infection and cancer. A major mechanism of immunity in human cells is the cGAS-STING nucleotide signaling pathway that responds to DNA aberrantly localized in the cytosol during cancer, cellular stress, and pathogen infection. Upon binding to cytosolic DNA, the enzyme cGAS catalyzes formation of cyclic GMP–AMP (2′3′-cGAMP), a nucleotide immune signal that activates the receptor STING to initiate an immune gene expression program. Due to broad tissue tropism and the ability to potently respond to natural small-molecules, cGAS-STING signaling is an emerging target for cancer immunotherapy. Our research demonstrates that cGAS is part of a large family of signaling enzymes that includes animal pattern recognition receptors named cGAS-Like Receptors (cGLRs) and bacterial anti-phage defense proteins named cGAS/DncV-like nucleotidyltransferase (CD-NTase) enzymes. Using structural and biochemical approaches, we are working to discover how nucleotide immune signals control immunity in all kingdoms of life:
How do new classes of nucleotide immune signals control human and bacterial immune systems?
How do cGLRs and novel signaling enzymes shape the cellular response to viral infection and disease?
Evolution of Antiviral Immunity
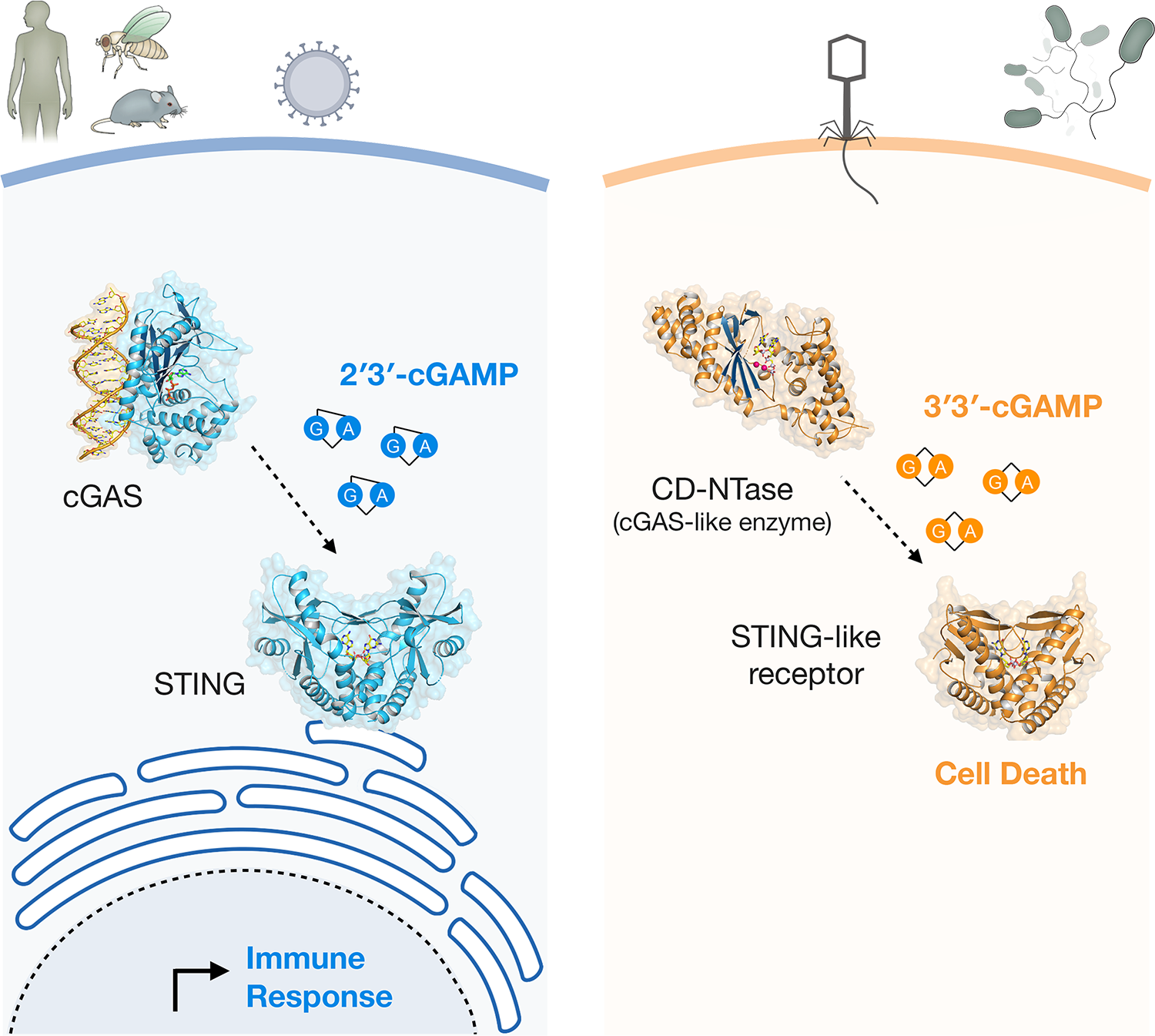
Our research reveals the surprising discovery that human innate immunity evolved from ancient pathways in bacteria. Combining structural biology, cell biology, and novel forward biochemical screening approaches, our work explains the mechanism of how human cells recognize infection and demonstrates that the core components that control these signaling pathways are descended from proteins in bacteria responsible for anti-phage defense. The discovery of the ancient origins of antiviral immunity provides a new framework to explain organization of the human immune system and to define novel components that inhibit pathogen replication in animal cells. We are continuing to build on the discovery of the ancient origins of human immunity with the major goals to:
Discover new pathways that control the host response to infection.
Determine the structural basis of how bacterial and metazoan receptors sense viral replication.
Define the rules that explain how human viruses and bacteriophages defeat host immunity.